- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
What Is Alkaptonuria? The Rare Disease That Turns Your Pee Black – Here’s Why

Image Credit: Canva
A condition, known commonly as "black urine disease" or Alkaptonuria is a rare genetic disorder involving protein metabolism, and it has its root in the mutation of the homogentisate 1,2-dioxygenase gene, which in turn causes homogentisic acid accumulation in the body. The appearance of dark urine after exposure to air is due to this kind of accumulation; however, a variety of symptoms can be expected, such as joint stiffness, changes in pigmentation, and other long-term health complications. Although the prevalence has been estimated to be between 1 in 250,000 and 1 in 1 million people in the United States, its effects are indeed high on those affected.
Alkaptonuria is an autosomal recessive disease, meaning that the child must inherit a defective copy of the HGD gene from both parents. If both parents are carriers, their offspring have a 25% chance of inheriting two faulty genes and developing alkaptonuria. The condition is genetic but is often not diagnosed for years because it progresses slowly and its early symptoms appear to be harmless.
Symptoms of Alkaptonuria
The most characteristic and common initial symptom of alkaptonuria is dark urine. The reason for this is due to the fact that excess HGA is excreted in the urine and upon oxidation in the presence of air, it gives the urine a brown or black color. Though it is often considered cosmetic, the long-term accumulation of HGA within the connective tissues produces more complicated health problems.
Progressive joint pain and stiffness: The accumulation of HGA in cartilage leads to early-onset osteoarthritis, making movement increasingly difficult over time.
Skin and eye pigmentation changes: Affected individuals may develop bluish or grayish discoloration of the sclera (white part of the eye) and the skin, particularly in areas exposed to friction.
Cardiovascular and respiratory problems: With age, HGA accumulation can lead to valve calcifications in the heart and stiffening of connective tissues in the respiratory tract, which can cause problems in middle and old age.
Decreased mobility and spinal problems: The spine may become stiff and painful due to chronic cartilage degeneration.
These symptoms usually begin to manifest during adulthood, leading to severe complications in a person's 40s or 50s and significantly affecting the quality of their life.
How is Alkaptonuria Diagnosed?
Because of its rarity, alkaptonuria is often mistaken or overlooked early in life. However, there are several ways to confirm the condition:
Urine Testing: The gold standard in the diagnosis is the testing of urine samples for high levels of homogentisic acid via gas chromatography. In case of oxidation, which changes the color of urine to black, it is indicative of alkaptonuria.
Genetic Testing: Confirmatory genetic testing reveals mutations of the HGD gene to diagnose the condition conclusively.
Blood Tests: High levels of HGA in the blood can be used as further evidence.
Imaging Studies: X-rays and MRIs will expose cartilage and joint damage characteristic of alkaptonuria.
Management of Alkaptonuria: Is There A Cure?
At present, there is no cure for alkaptonuria; however, various treatment approaches can reduce its symptoms and slow the disease's progress:
Nitisinone Therapy: Nitisinone is a drug that inhibits the production of HGA. It has been shown to reduce HGA levels and slow tissue damage. However, it needs to be taken under close medical supervision because of potential side effects.
Low-Protein Diet: Since HGA is a byproduct of protein metabolism, reducing protein intake—especially foods rich in tyrosine and phenylalanine—may help decrease HGA production.
Pain Management: OTC pain relievers and anti-inflammatory medications can be used to relieve joint pain and stiffness.
Physical Therapy: Exercise regularly, as it may improve mobility and strengthen muscles, thus reducing strain on affected joints.
Surgical Interventions: Most people with alkaptonuria develop severe osteoarthritis necessitating joint replacement in their old age. Also, some may require heart valve replacement surgery if cardiovascular complications develop.
Life with Alkaptonuria
Although alkaptonuria is not fatal, it severely affects the quality of life. The progressive deterioration of the joints and associated symptoms can make everyday activities difficult, requiring lifestyle changes and medical interventions. The disease may cause premature aging of the joints, requiring walking aids and mobility assistance earlier than expected.
Ongoing research will continue to work on improving the treatment options by focusing on gene therapy and alternative enzyme replacement therapies. However, because of its rarity, the clinical trials and research remain sparse.
As genetic research advances, more hope for better management and possible curative approaches for alkaptonuria exists. Scientists are searching extensively for enzyme replacement therapies and innovative drugs that can target the root cause of the disorder. Being aware and being diagnosed early helps individuals better their condition and ultimately have better long-term health outcomes.
Alkaptonuria is a striking example of how one gene mutation can have widespread effects on the body. Though still a rare and often misunderstood condition, growing awareness and advances in treatment are paving the way for better care. If you or a loved one suspect symptoms of alkaptonuria, it is essential to seek early diagnosis and medical guidance to manage the disease effectively and preserve quality of life.
Can An App Be Your Therapist? India’s Digital Mental Health Revolution Faces Key Challenges

Credit: iStock
Millions of Indians are increasingly turning to mental health apps to manage stress, anxiety, loneliness, and other emotional challenges. These digital platforms offer a range of services, from mood tracking and mindfulness exercises to AI-powered chatbots designed to provide conversational support that can feel similar to therapy.
The growing adoption of such apps comes amid a significant shortage of mental health professionals in India. Data from the National Mental Health Survey indicate that nearly one in ten Indian adults lives with a diagnosable mental health disorder, while more than 85 percent of people with depression do not receive adequate treatment. Other studies have also highlighted a large gap between the demand for mental healthcare services and the availability of trained psychiatrists, psychologists, and counselors, particularly in rural and underserved regions.
Against this backdrop, mental health apps position themselves as a convenient and relatively affordable way to access support anytime and from anywhere. However, researchers and mental health experts caution that the rapid rise of these platforms has also sparked concerns about data privacy, the clinical effectiveness of digital interventions, and the level of oversight and accountability surrounding AI-driven mental health tools. While some studies suggest that digital mental health programs can help reduce symptoms of anxiety and depression, experts emphasize that they should complement—not replace—professional care, especially for individuals with moderate to severe mental health conditions.
Also Read: India's Maternal Mortality Ratio Drops From 130 To 87 In Last 10 Years: Govt
How Mental Disorders Are Impacting Life Worldwide?
Mental disorders are on the rise worldwide, and nearly 1.2 billion lives are now affected by them. A recent study published in The Lancet has revealed this very crucial trend. The study was conducted by researchers at the Institute for Health Metrics and Evaluation (IHME) in collaboration with the University of Queensland.
This study shows that the number of people affected by the mental disorder has almost doubled since 1990. It also emphasizes the role of mental disorder in the case of disability, as the research suggests that it has crossed cardiovascular disease, cancer, and musculoskeletal conditions, leading to disability. Notably, the study has revealed that mental disorders affect people of different sexes and ages disproportionately.
The research was done on a massive scale from 1990 to 2023. The researchers studied the impact of mental disorder on both sexes, in 25 age groups, 21 regions, and 204 countries and territories.
Over the years, mental disorders have caused more and more mayhem in people's lives. Disability adjusted life years (DALYs) show the very impact of mental disorders on life. DALYs measure the health loss. It calculates the years lived with disability and years of life lost due to premature death.
According to this very measuring system, we can see that the mental disorder and its impact on overall life reached another zenith in 2023. That year, mental disorders alone caused 171 million disability adjusted life years (DALYs). This made mental disorder the fifth biggest contributor to total disease burden. Notably, in 2023, mental disorders had a 17 percent share of total life lost due to disability throughout the world.
Bleeding Disorders In Women: Gynaecologist Lists The Causes
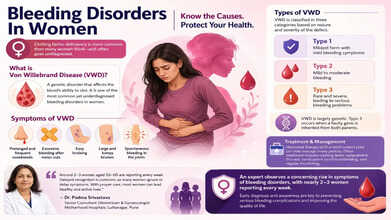
Early diagnosis and awareness are key to preventing serious bleeding complications and improving the quality of life. (Photo credit: AI generated)
Currently, there is a spike in cases of women being diagnosed with clotting factor deficiency, a condition where the blood does not clot properly. Many women aren’t aware of this and tend to dismiss it as routine menstrual concerns. While not always widely discussed, this condition can lead to serious health risks if not identified early. Hence, women should be more attentive and consult the doctor when they notice unusual bleeding patterns and improve their overall well-being.
What is Von Willebrand Disease?
One of the most common yet underdiagnosed conditions in women is von Willebrand disease. So, it is a genetic disorder that affects the blood’s ability to clot. Some women present as carriers of hemophilia or develop clotting issues due to liver disease, infections, hormonal imbalance, or nutritional deficiencies such as low vitamin K. Hence, women should pay utmost attention to their health.
What are the symptoms of VWD?
VWD is a common inherited bleeding disorder, and it occurs when there is a dysfunctional blood protein that does not let platelets clump together to form clots. Because it adversely affects the blood's ability to clot, it causes mucosal bleeding. In mild cases, the symptoms may not be there, but in other cases, the symptoms are the following:
- Prolonged and frequent nosebleeds
- Excessive bleeding after minor cuts
- Easy bruising
- Large and lumpy bruises
- Spontaneous bleeding in the joints
What are the types of VWD?
VWD is classified in three categories based on the nature and severity of the defect:
- Type 1: Mildest form with mild bleeding symptoms
- Type 2: Mild to moderate bleeding
- Type 3: Rare and severe, leading to serious bleeding problems
VWD is largely genetic, and the most extreme form—type 3—occurs when a faulty gene is inherited from both parents. For treatment, doctors recommend hormonal therapy like IUDs or birth control pills to manage heavy period bleeding.
“Clotting factor deficiency is seen when the body lacks certain proteins needed to stop bleeding. In women, this may be a genetic condition, such as hemophilia carriers or other inherited bleeding disorders. They can experience clotting factor deficiency because of liver problems, infections, hormonal imbalances, or nutritional deficiencies like low vitamin K levels. Women avoid the symptoms like bleeding gums, heavy period bleeding, and other issues, assuming that the symptoms with get better. However, prolonged bleeding is also reported by women, especially after gynaecological procedures like childbirth," Dr. Padma Srivastava, Senior Consultant Obstetrician & Gynaecologist, Motherhood Hospitals, Lullanagar, Pune, said in an interview with Health and Me.
Dr. Padma further added, “Around 2–3 women aged 50-60 are reporting every week. However, delayed recognition remains a major challenge, as many women tend to ignore symptoms or delay seeking medical advice. Management of this condition involves clotting factor replacement therapy, medications to control bleeding, and regular monitoring. With proper care, most women can lead healthy and active lives.”
Pregnancy After 35: Is It Prone To Complications?

Going for timely fertility evaluations is a good way to dodge the risk of pregnancy complications. (Photo credit: AI generated)
Changing lifestyles, career priorities, financial planning, and personal choices are leading many couples to marry later than before. While late marriages are becoming increasingly common, fertility specialists are observing a parallel rise in delayed pregnancy planning and age-related fertility complications. Many couples now begin trying to conceive only after the age of 35, when fertility in both men and women naturally starts declining.
Currently, there is a growing trend of late marriages due to career priorities, financial stability goals, higher education, and changing lifestyle choices. Many couples are choosing to settle professionally and personally before planning a family. However, this delay can also lead to postponed pregnancy planning, often at a stage when fertility naturally starts declining. Increasing stress, underlying medical conditions, and lack of fertility awareness are further contributing to challenges in conception among couples in their mid to late thirties.
What are the complications that can occur in a pregnancy after turning 35?
“The increasing trend of late marriages (30–35+) is directly leading to delayed pregnancy planning. Most couples start trying to conceive after 35, when fertility has already begun to decline significantly. By the time many patients seek medical help between 36 and 39 years, several are diagnosed with low ovarian reserve (low AMH) or conditions such as oligoasthenoteratozoospermia (OAT) and azoospermia.
Lack of early fertility awareness often causes women to miss the window for timely intervention or fertility preservation options like egg freezing. Advanced maternal age pregnancies (40+) are steadily rising and are often associated with complex complications and lower treatment success rates. Currently, nearly 75% of fertility patients are above 35 years of age, while 25% are above 40. Only around 20% of patients are in their late 20s,” said Dr. Jyotshana Palgamkar, Fertility Specialist, Nova IVF Fertility, Virar.
Dr Jyotshna further added, “One of the biggest concerns linked to late pregnancy planning is reduced ovarian reserve in women. By the time many women seek medical help between the ages of 36 and 39, their anti-Müllerian hormone (AMH) levels, which indicate egg reserve, may already be low. This reduces the chances of natural conception and can also affect the success rates of fertility treatments. Male fertility is also being affected. A large number of men are diagnosed with oligoasthenoteratozoospermia (OAT), a condition in which sperm count, motility, and morphology are affected.
Consequences of delayed fertility evaluation
Delayed fertility evaluation often means that couples lose valuable time before beginning treatment or considering assisted reproductive options. Another major challenge is the lack of awareness about fertility preservation. Many women remain unaware that egg freezing at a younger age may help preserve fertility for the future. By the time they start exploring fertility options in their late thirties or forties, treatment becomes more complex and emotionally stressful. Advanced maternal age pregnancies, especially above 40, are also increasing. These pregnancies are often associated with lower conception rates, higher miscarriage risk, pregnancy complications, and reduced IVF success rates,” Dr Jyotshna explained.
Male fertility treatment options depend on the underlying cause of infertility and may include lifestyle changes, medications, hormonal therapy, or surgical procedures. “Conditions such as low sperm count, poor sperm motility, abnormal morphology, raised DNA fragmentation index (DFI) affecting sperm quality, varicocele, or azoospermia can often be managed with timely medical intervention. Assisted reproductive techniques like IUI, IVF, and ICSI are also commonly used to improve the chances of conception. Early diagnosis and proper fertility evaluation play an important role in selecting the most effective treatment option for men,” said Dr Palgamkar.
Understanding fertility timelines, seeking timely evaluation, and discussing preservation options early can help couples make informed decisions and improve their chances of successful parenthood in the future.
© 2024 Bennett, Coleman & Company Limited

