- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
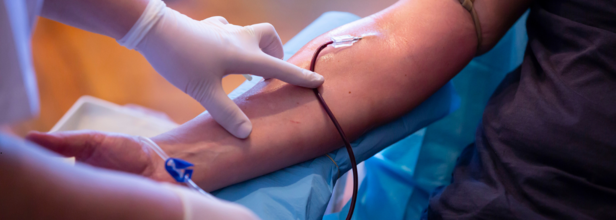
How Long After a Tattoo or Piercing Can I Donate Blood?
Credits: Canva
Are you that kind of person who celebrates milestones of your life with getting a tattoo? These milestones could be anything, including the things you achieved, or the things you could not achieve but taught you a lesson. If you are this person, then you must have wondered if you can donate blood with all the tattoos on your body? There are lots of rumors on how can one donate blood, or if at all they are allowed to donate blood. So let's get into its nitty gritty!
As per American Red Cross, in most states, a tattoo is acceptable if the tattoo was applied by a state-regulated entity. Which means the tattoo artist must be licensed and must practice following all the guidelines, using sterile needles and ink that is not reused. The same is the guideline for cosmetic tattoos, which includes microblading of eyebrows. If it is done by a licensed artist in a regulated state, then it is acceptable.
However, if you got your tattoo in a state that does not regulate tattoo facilities, you must wait three months after it was applied.
The states that do not regulate tattoo facilities are:
- Arizona
- District of Columbia
- Georgia
- Idaho
- Maryland
- Massachusetts
- Nevada
- New Hampshire
- New York
- Pennsylvania
- Utah
- Wyoming
Body Piercing
Similar is the case with body piercings. It has to be done following the regulation, here the key is that the instrument used has to be a single-use equipment and disposable. Which means if you are getting it by a gun, or an earring cassette, they have to be disposable. In case you got your piercing with a reusable gun or a reusable instrument, you will be required to wait for three months.
Three-Month Wait Period
The reason behind the wait time is associated with the concerns of hepatitis, which could be easily transmitted from donors to patients through transfusion. All blood donations are thus tested for hepatitis B and hepatitis C, with several tests. However, not always are these tests are perfect, thus the three-month period is given.
What Dangers Loom Over?
Donating blood after getting a tattoo can be dangerous as unclean tattoo needle could carry bloodborne viruses, which are hepatitis B, hepatitis C and HIV. In 2020, the Food and Drug Administration (FDA) updated its guideline, making the wait time shorter from one year to three months. This is because if you contract a bloodborne illness, it could be detectable within the period of 3 months.
What else makes you ineligible to donate blood?
There are other reasons why you may not be allowed to donate blood. As per the American Red Cross, you are not allowed to donate blood if you have
- hepatitis B or C
- HIV
- Chagas disease, which is a parasitic infection that kissing bugs cause
- leishmaniasis, a parasitic infection that sand flies cause
- Cruetzfeldt-Jakob Disease (CJD), a rare disorder that leads to mental deterioration
- Ebola virus
- hemochromatosis, which means extreme build up of iron
- hemophilia
- jaundice
- sickle cell disease
As per the National Institutes of Health (NIH) Blood Bank, these conditions make you permanently ineligible from donating blood.
While there are certain conditions that makes your permanently ineligible, there are other conditions that makes you temporarily ineligible from donating blood. These include:
- If you have a bleeding condition, and have issues with your blood clotting
- If you have received transfusion from a person
- If you have cancer. Here, the eligibility depend son the type of cancer you have
- If you have recently underwent a dental or oral surgery. In such a case, you would have to wait for three days
- If you had a recent heart attack, heart surgery or angina. You must wait for 6 months
- If you are pregnant, you can only donate blood after 6 months after delivering your child
New Drug May Prevent Muscle Loss Linked To Ozempic And Weight-Loss Shots

Credit: AI Generated Image
A recent research published in the Nature Medicine journal reveals that a new drug can help people on obesity shots to avoid unwanted muscle loss linked to flat bums or "Ozempic butt". The US research suggests that about a third of the weight loss from GLP-1 obesity jabs like Wegovy and Mounjaro can come from muscle rather than fat. The medication is apitegromab.
The trial with 102 adults, mostly women, who took apitegromab with their obesity medication, maintained more muscle while still losing fat, body scans showed.
But How Does Ozempic Function?
The first thing to remember here is that Ozempic is a brand-name medicine that contains semaglutide as its active ingredient. Semglutide is the synthetic version of GLP-1, a natural hormone produced in the intestines that regulates blood sugar, appetite, and digestion. Now, every time you eat, your body produces various hormones, including GLP-1. These are called post-nutrition hormones, and they help you absorb the energy you just consumed.
Also Read: Salmonella Outbreak: Instant Noodles Sicken Over 80 In The UK, Europe
GLP-1 travels to your pancreas, prompting it to produce insulin. It also travels to the hypothalamus in your brain, which gives you the feeling of being full or satiated. Ozempic imitates this hormone, thereby silencing the food chatter in the brain. Interestingly, for some people, this food chatter is really quiet ( people with low appetite), and for others it is an outburst (people who generally binge eat). So with Ozempic, silencing this self-talk in the brain, people tend to lose their appetite and eventually weight.
However, it is important to note that losing weight includes not just fat but muscle as well. Losing too much muscle can lead to reduced strength and a shorter life span. Notably, records show that most people who start taking them stop them at 12 weeks; therefore, it is important for some but not for others.
Also Read: Ebola Outbreak Escalates: Uganda Rushes Medics To Congo Border Regions
What Are The Side Effects?
Ozempic is a drug that is tasked to help diabetic patients manage their blood sugar levels and weight. However, recent research has shown its effectiveness in mitigating various addictions like alcohol and drugs by inhibiting hormones. But what people ignore are its side effects, which include:
- Nausea is a frequent side effect, especially when starting Ozempic or increasing the dose, and vomiting may occur along with nausea.
- Diarrhoea and abdominal discomfort also show up in people using Ozempic, but they generally resolve as your body adjusts.
- Ozempic can reduce appetite but may also lead to unintended weight loss or reduced food intake, causing discomfort for some people.
- There are certain less common, but serious side effects, like Pancreatitis, or inflammation of the pancreas.
- This drug may also cause severe kidney issues, particularly if dehydration occurs from side effects like vomiting or diarrhoea.
Can Young Adults Get Brain Tumours?

Young adults must go for regular health check-ups, especially in case of persistent headaches. (Photo credit: AI generated)
Although brain tumours are more commonly associated with older adults, they can occur at any age, including during adolescence and young adulthood. Recognising the warning signs early can help ensure timely diagnosis and treatment. Here, an expert explains the symptoms and highlights when medical attention should be sought.
What is a brain tumour?
In an interview with Health and Me, Dr Rakesh Patil, Consultant Medical Oncologist, AIMS Hospital, Dombivli, spoke about brain tumours and their rising incidence among younger Indians.
Brain tumours occur when abnormal cells grow uncontrollably within the brain or surrounding tissues. These tumours may be benign (non-cancerous) or malignant (cancerous). While brain tumours are not among the most common cancers affecting young adults, cases do occur in this age group. The exact cause of most brain tumours remains unknown. However, certain genetic conditions, a family history of brain tumours, previous exposure to radiation therapy, and rare inherited disorders may increase the risk. In many cases, affected individuals have no identifiable risk factors. Early diagnosis and appropriate treatment remain crucial for achieving the best possible outcomes.
Why are brain tumour symptoms missed?
One of the major challenges is that the early symptoms of a brain tumour can be mistaken for stress, migraines, sleep deprivation, or eye strain. Persistent headaches, especially those that worsen over time; nausea; vomiting; blurred or double vision; balance problems; unexplained seizures; weakness in an arm or leg; personality changes; memory difficulties; and problems with speech or concentration should not be ignored and warrant prompt medical evaluation.
Why are brain tumour cases rising among young Indians?
Many young adults are occupied with studies, careers, and social commitments, which may lead them to overlook symptoms or delay seeking medical attention. If left untreated, a brain tumour can affect important brain functions, including movement, speech, vision, memory, and behaviour. In some cases, it can become life-threatening.
What are the best treatment options for brain tumour?
Advances in imaging technology, neurosurgery, radiation therapy, and targeted treatments have significantly improved outcomes for many patients. Following a thorough evaluation and diagnosis, the treating physician will determine the most appropriate treatment plan based on the type, size, and location of the tumour. Some patients may also benefit from neurorehabilitation as part of their recovery.
Young adults should seek medical evaluation if they experience persistent headaches, develop neurological symptoms, or have seizures without an obvious cause. Greater awareness and timely medical attention can help improve outcomes and, in some cases, save lives.
AIIMS Delhi Study Warns Air Pollution May Harm Unborn Babies

Credits: iStock
Air pollution is one of the most impactful issues for the people of Delhi, as the city is known for having heavy air pollution, and the whole NCR faces this particular health hazard due to very high PM2.5 concentrations.
Though until now there was a public perception that the effect of this situation on babies was not massive, but that hopeful thinking has been shattered by a new study done by the All India Institute of Medical Sciences (AIIMS), Delhi.
The study by AIIMS has revealed that when fine particulate matter is found in the air, it can damage the placenta, restrict fetal growth, and increase the risk of low birth weight.
It was published in EMBO Molecular Medicine and focused on the effect of fine particulate matter in the air on pregnancy through laboratory study, animal models, and human data.
The research team observed that the presence of fine particulate matter in the air causes harm to placental function, including blood vessel formation, nutrient transport, and fetal growth.
In animal trials, the team has observed that the rats exposed to pollution have smaller litters, abnormal placentas, and babies with reduced birth weight and growth.
The research was supported by an analysis of 994 pregnancies from regions with different pollution levels. The team of AIIMS also stated that they have seen PM2.5 exposure was associated with a greater risk of low birth weight and preeclampsia.
How To Reduce Climate-related Health Risks
To reduce climate-related health risks, the experts recommend staying informed about weather conditions, remaining hydrated, limiting outdoor exposure during extreme heat, and taking precautions against mosquito bites.
People with chronic conditions such as hypertension, diabetes, heart disease, and kidney disease are advised to maintain adequate medication supplies and consult healthcare providers during periods of extreme heat.
Also Read: Meningitis Alert at UK University: Preventive Antibiotics and Vaccination Offered to Students
According to the experts, awareness and early action remain critical to protecting public health as climate change continues to reshape disease patterns around the world.
To reduce the risk of mosquito-borne diseases, people should make sure that water does not get collected in buckets, coolers, flower pots, or other containers around the home. Additional precautions include the use of mosquito repellents, sleeping under nets when necessary, and wearing clothing that covers the arms and legs.
Dr Sabine said food and water safety are particularly important after heavy rain or flooding. Proper food storage, clean drinking water, and good hygiene practices can go a long way in preventing many infections. Those with pre-existing medical conditions should also have essential medicines within easy reach during extreme weather.
© 2024 Bennett, Coleman & Company Limited

