- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Can Damaged Sperm Lead To Pregnancy Complications? Study Highlights Uncommon Issues
Can Damaged Sperm Lead To Pregnancy Complications?
Pregnancy is usually a time of happiness and hope, but it also brings in the unexpected. While there is much talk placed on the health of the expectant mother, the quality sperm coming from the father could dramatically change the outcome of pregnancy. The latest study suggests the risks of sperm DNA damage, even increasing the risks of complications preeclampsia and birth prematurity.
In a groundbreaking research study conducted by scientists from Lund University in Sweden, scientists discovered that DNA damage in sperm increases the risk almost up to double that of preeclampsia, this is a dangerous condition that may arise during pregnancies characterized by high blood pressure. In addition, DNA anomalies also increase the risk of premature births, and this further entails increased related adverse health outcomes for infants born through such conditions.
The next step would be to find out which group of men respond best to methods to prevent and treat sperm DNA damage, and to test these methods to prevent pregnancy complications," said Dr. Amelie Stenqvist, a lecturer at Lund University. According to this study, a significant message is put forward that paternal health assumes an important role in a successful pregnancy.
It focused its research on men, specifically whose sperm contained high levels of DNA fragmentation. For instance, some 20% to 30% of babies born via in vitro fertilization have fathers whose sperm contains damaged DNA. The DNA fragmentation index, an indicator to assess the percentage of DNA damage in sperm, indicated that when the percentage of sperm with a DFI above 30% was observed, they had almost no chance of resulting in natural conception. Even a DFI greater than 20% showed that the chances of getting pregnant are highly risky as the risk factor for pregnancy complications like preeclampsia is much high.
Uncommon Complications during Pregnancy
The most alarming complication during pregnancy is preeclampsia. It affects approximately 5% to 8% of pregnancies worldwide, which can cause fatal conditions for both the mother and the baby. The new findings now point out that sperm DNA damage may contribute to this condition, especially if it is due to assisted reproductive techniques such as IVF pregnancies. The research found that a DFI above 20% doubled the risk of preeclampsia from a mere 5% to almost 11% per.
Apart from causing preeclampsia, DNA fragmentation in sperm is also known to increase the risk for prematurity. Most premature babies experience respiratory, neurological, and developmental complications. Therefore, some degree of early intervention might be important for prospective parents.
Some of the rarer, though serious complications include placental abruption, which is the separation of the placenta from the uterine wall and intrauterine growth restriction, a condition by which the baby does not grow normally in the womb. These conditions though rare are potentially catastrophic both to the mother and the child. Results from this study may help in establishing the contribution of the father in such pregnancies.
Further study into sperm DNA damage is of urgent interest with regard to its consequences for pregnancy outcomes. According to Professor Aleksander Giwercman of Lund University in the field of Reproductive Medicine, "the analysis of DFI should be introduced as routine test in all fertility clinics.". "It could give answers to couples who are having difficulties with infertility, but our latest result also shows that DFI analysis can be a method to identify high-risk pregnancies, explained Giwercman.
For many, DNA fragmentation in sperm is often treatable. Common causes are oxidative stress, age, smoking, being obese, and infections. Addressing these elements will likely reduce DNA damage in sperm for men, raising the chances for a healthy pregnancy and baby.
Overall, the study importance should take into consideration paternal as well as maternal health towards reaching for a healthy pregnancy. Though DNA fragmentation in the sperm is supposed to increase the risk factors for complications in pregnancies, the advances into novel treatment approaches and tests are likely to alleviate complications in many families. Thus the findings of this study offer optimism and pave a pathway to more holistic fertility treatments in the future.
Bleeding Disorders In Women: Gynaecologist Lists The Causes
Early diagnosis and awareness are key to preventing serious bleeding complications and improving the quality of life. (Photo credit: AI generated)
Currently, there is a spike in cases of women being diagnosed with clotting factor deficiency, a condition where the blood does not clot properly. Many women aren’t aware of this and tend to dismiss it as routine menstrual concerns. While not always widely discussed, this condition can lead to serious health risks if not identified early. Hence, women should be more attentive and consult the doctor when they notice unusual bleeding patterns and improve their overall well-being.
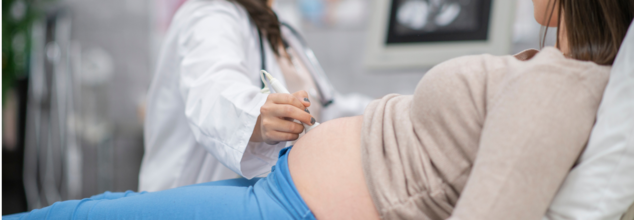
What is Von Willebrand Disease?
One of the most common yet underdiagnosed conditions in women is von Willebrand disease. So, it is a genetic disorder that affects the blood’s ability to clot. Some women present as carriers of hemophilia or develop clotting issues due to liver disease, infections, hormonal imbalance, or nutritional deficiencies such as low vitamin K. Hence, women should pay utmost attention to their health.
What are the symptoms of VWD?
VWD is a common inherited bleeding disorder, and it occurs when there is a dysfunctional blood protein that does not let platelets clump together to form clots. Because it adversely affects the blood's ability to clot, it causes mucosal bleeding. In mild cases, the symptoms may not be there, but in other cases, the symptoms are the following:
- Prolonged and frequent nosebleeds
- Excessive bleeding after minor cuts
- Easy bruising
- Large and lumpy bruises
- Spontaneous bleeding in the joints
What are the types of VWD?
VWD is classified in three categories based on the nature and severity of the defect:
- Type 1: Mildest form with mild bleeding symptoms
- Type 2: Mild to moderate bleeding
- Type 3: Rare and severe, leading to serious bleeding problems
VWD is largely genetic, and the most extreme form—type 3—occurs when a faulty gene is inherited from both parents. For treatment, doctors recommend hormonal therapy like IUDs or birth control pills to manage heavy period bleeding.
“Clotting factor deficiency is seen when the body lacks certain proteins needed to stop bleeding. In women, this may be a genetic condition, such as hemophilia carriers or other inherited bleeding disorders. They can experience clotting factor deficiency because of liver problems, infections, hormonal imbalances, or nutritional deficiencies like low vitamin K levels. Women avoid the symptoms like bleeding gums, heavy period bleeding, and other issues, assuming that the symptoms with get better. However, prolonged bleeding is also reported by women, especially after gynaecological procedures like childbirth," Dr. Padma Srivastava, Senior Consultant Obstetrician & Gynaecologist, Motherhood Hospitals, Lullanagar, Pune, said in an interview with Health and Me.
Dr. Padma further added, “Around 2–3 women aged 50-60 are reporting every week. However, delayed recognition remains a major challenge, as many women tend to ignore symptoms or delay seeking medical advice. Management of this condition involves clotting factor replacement therapy, medications to control bleeding, and regular monitoring. With proper care, most women can lead healthy and active lives.”
Pregnancy After 35: Is It Prone To Complications?

Going for timely fertility evaluations is a good way to dodge the risk of pregnancy complications. (Photo credit: AI generated)
Changing lifestyles, career priorities, financial planning, and personal choices are leading many couples to marry later than before. While late marriages are becoming increasingly common, fertility specialists are observing a parallel rise in delayed pregnancy planning and age-related fertility complications. Many couples now begin trying to conceive only after the age of 35, when fertility in both men and women naturally starts declining.
Currently, there is a growing trend of late marriages due to career priorities, financial stability goals, higher education, and changing lifestyle choices. Many couples are choosing to settle professionally and personally before planning a family. However, this delay can also lead to postponed pregnancy planning, often at a stage when fertility naturally starts declining. Increasing stress, underlying medical conditions, and lack of fertility awareness are further contributing to challenges in conception among couples in their mid to late thirties.
What are the complications that can occur in a pregnancy after turning 35?
“The increasing trend of late marriages (30–35+) is directly leading to delayed pregnancy planning. Most couples start trying to conceive after 35, when fertility has already begun to decline significantly. By the time many patients seek medical help between 36 and 39 years, several are diagnosed with low ovarian reserve (low AMH) or conditions such as oligoasthenoteratozoospermia (OAT) and azoospermia.
Lack of early fertility awareness often causes women to miss the window for timely intervention or fertility preservation options like egg freezing. Advanced maternal age pregnancies (40+) are steadily rising and are often associated with complex complications and lower treatment success rates. Currently, nearly 75% of fertility patients are above 35 years of age, while 25% are above 40. Only around 20% of patients are in their late 20s,” said Dr. Jyotshana Palgamkar, Fertility Specialist, Nova IVF Fertility, Virar.
Dr Jyotshna further added, “One of the biggest concerns linked to late pregnancy planning is reduced ovarian reserve in women. By the time many women seek medical help between the ages of 36 and 39, their anti-Müllerian hormone (AMH) levels, which indicate egg reserve, may already be low. This reduces the chances of natural conception and can also affect the success rates of fertility treatments. Male fertility is also being affected. A large number of men are diagnosed with oligoasthenoteratozoospermia (OAT), a condition in which sperm count, motility, and morphology are affected.
Consequences of delayed fertility evaluation
Delayed fertility evaluation often means that couples lose valuable time before beginning treatment or considering assisted reproductive options. Another major challenge is the lack of awareness about fertility preservation. Many women remain unaware that egg freezing at a younger age may help preserve fertility for the future. By the time they start exploring fertility options in their late thirties or forties, treatment becomes more complex and emotionally stressful. Advanced maternal age pregnancies, especially above 40, are also increasing. These pregnancies are often associated with lower conception rates, higher miscarriage risk, pregnancy complications, and reduced IVF success rates,” Dr Jyotshna explained.
Male fertility treatment options depend on the underlying cause of infertility and may include lifestyle changes, medications, hormonal therapy, or surgical procedures. “Conditions such as low sperm count, poor sperm motility, abnormal morphology, raised DNA fragmentation index (DFI) affecting sperm quality, varicocele, or azoospermia can often be managed with timely medical intervention. Assisted reproductive techniques like IUI, IVF, and ICSI are also commonly used to improve the chances of conception. Early diagnosis and proper fertility evaluation play an important role in selecting the most effective treatment option for men,” said Dr Palgamkar.
Understanding fertility timelines, seeking timely evaluation, and discussing preservation options early can help couples make informed decisions and improve their chances of successful parenthood in the future.
Poor Posture, Disc Bulge: What Causes Back Pain In Young People
Lack of exercise can contribute to back pain. (Photo credit: AI generated)
Back pain is generally considered a problem seen in old age, but of late, we are increasingly seeing younger adults suffering from back pain. Prolonged sitting hours, inadequate physical activity, smoking, and an unhealthy lifestyle have made back pain a common complaint even in people under 30 years of age.
What is a slipped disc?
In an interview with Health and Me, Dr Pramod Sudarshan, Spine Surgeon (ortho), Apollo Hospitals, Bangalore, said, “Most commonly, we attribute this pain to a slipped disc or other disc-related conditions. However, they are just one among many reasons causing back pain. A thorough understanding of the other conditions that can lead to back pain, and knowing the ‘red flags’ to help identify them, will assist in seeking early treatment and preventing further complications.”
Muscle strain is considered to be one of the most common reasons for developing back pain in younger individuals. Daily activities like travelling for long hours, lifting heavy weights without proper technique, and carrying heavy backpacks can lead to muscle strain. Often, the pain due to muscle strain comes down with the help of medications such as muscle relaxants and rest.
Poor posture and back pain
Working professionals and students spend many hours a day studying, gaming, and working on computers without maintaining proper posture, leading to excess strain on the back muscles and, in turn, prolonged back pain. Having a properly ergonomically designed workstation/study table setup can prevent back discomfort and stiffness.
What are the causes of back pain?
Lack of physical activity and associated conditions such as obesity weaken our core muscles, which are considered the main pillars supporting our spine. Weakened core muscles will not be able to perform the functions expected of them, leading to back pain. Regular exercise, weight management, a healthy diet, and guided weight training will help strengthen our core muscles and reduce the risk of back pain.
Other than muscles, the spine also contains small joints known as facet joints, irritation or inflammation of which can lead to back pain. A structure known as the pars interarticularis acts as a clamp in the spine, holding two adjacent vertebral bodies, and any defect, such as a congenital defect or fracture due to repetitive microtrauma, as seen in gymnasts, can lead to the slipping of one bone over the other, causing back pain.
Though mechanical back pain is most common, there are other conditions such as inflammatory arthropathy, tumours, and infections. Conditions such as ankylosing spondylitis, rheumatoid arthritis, and hyperuricemia/gout are some examples of inflammatory conditions in which an individual experiences early morning back pain associated with stiffness and may also present with pain in other joints.
Can tuberculosis lead to back pain?
Tuberculosis affecting the spine is considered the most common infection of the spine. Severe back pain, weight loss, loss of appetite, fever, and night sweats are some of the common symptoms. Other bacterial and fungal infections are also seen affecting the spine, most commonly in immunocompromised individuals.
Tumours, though rare in young individuals, warrant further investigation when symptoms such as severe weight loss, night pain, and prolonged back pain are present to rule out the possibility of tumours.
Other conditions such as scoliosis (congenital/adolescent idiopathic scoliosis), which is a three-dimensional deformity of the spine, can cause back pain. It requires further assessment in the form of radiological investigations to assess the degree of deformity and to plan any surgical intervention, if required, based on the severity of the curve.
What are the symptoms?
Identifying some of the red-flag signs, such as weakness in the legs, numbness around the groin, bladder or bowel dysfunction, fever, unexplained significant weight loss, or severe pain following major trauma, is essential. These symptoms may indicate serious underlying pathology requiring medical care.
Fortunately, back pain in young people is largely preventable. Basic lifestyle modifications such as regular exercise, maintaining proper posture, avoiding prolonged sitting, maintaining a healthy body weight, undertaking monitored weight-training programmes, following a healthy diet, and getting adequate sleep will help prevent as well as overcome existing back problems. Invest in your spinal health at the earliest opportunity to reap the benefits in the second half of life.
© 2024 Bennett, Coleman & Company Limited

