- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
4 Ways You Can Treat Your Spring Allergies

Credits: Canva
Spring is around the corner, it means warmer weather, flowers blooming, but also pollen, allergy, lots of sneezing, wheezing and itchy eyes. If you are someone who rarely enjoys this season, and spends most of it battling congestion and brain fog, then this is for you! Learn how to manage your spring allergies.
Spring allergies can be treated with simple lifestyle changes, and if at all it is required, even advanced medical interventions too can take care of it. Here are some tips that the American College of Allergy, Asthma & Immunology (ACAAI) recommends:
Home Remedies
Before turning to medications, try considering these lifestyle changes and see if it works:
1. Shut Your Windows: Pollen counts are usually at the highest in early morning and evening. This is the time when you can start keeping your windows and doors closed. Whether you are at your home or in your car, this way you can prevent pollen from entering. You can also use air conditioning instead of opening the windows and keep the indoor cool.
2. Bed-Time Routine: If you have spent your day outdoors, the best thing to do is to take a shower and change your clothes before you hit the bed. This is a great way to get rid of pollen from your clothes, skin, and hair. This will reduce your chance of sleeping om a pillow full of pollen
3. Air Purifiers: You can use HEPA, also known as the High-efficiency particulate air filters, which can catch pollen, pet dander and dust and other kinds of air purifier to filter out allergens.
4. Limit Outdoor Activities: Try to limit your activities especially early in the morning and in the evening. If you must go, put on a mask. An N-95 mask, which had peaked during COVID-19 times could come handy even now.
5. Spring Cleaning: Clean your house, and other surfaces. Even if you do not see the dust, it is there, and cleaning those surfaces will help you get rid of accumulation of pollen and other allergens.
OTC Medications
In case these lifestyle changes are not helping you, you can go for over-the-counter medications. These include:
1. Antihistamines: They block histamine, which is a chemical released in body during an allergic reaction. It also reduces symptoms like sneezing, runny nose and itchy eyes.
2. Nasal Sprays: These could be your best friend if you are struggling with congestion. You can also use saline nasal sprays and neti pots.
3. Decongestants: These help by narrowing blood vessels in the nasal passages, and reduce swelling, which in return improves airflow.
Note that we do not recommend or prescribe any medicines. It is always advisable to go see your GP first.
Prescribed Medications
Your doctor is most likely to prescribe a desloratadine in case your symptoms are not controlled with OTC medications. You may also be prescribed azelastine that could work better on your inflammation and congestion.
If you have severe allergy symptoms, you may be put on corticosteroids for short-term oral use. It can reduce inflammation throughout the body.
Allergy Immunotherapy
This is called allergy immunotherapy, which is a long-term solution that can desensitize your immune system to specific allergens. This treatment is considered when other treatments are ineffective and when allergies significantly impact your daily life.
The two allergy immunotherapy involves, a shot called subcutaneous immunotherapy or SCIT, and sublingual immunotherapy or SPLI, which is an allergen tablets placed under the tongue.
How Liver Disease Affects The Heart
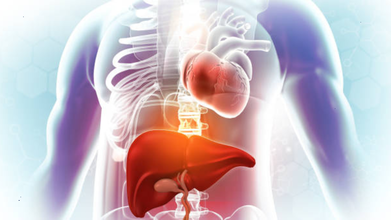
Credit: iStock
The liver and heart are two of the most vital organs of the body, and although they perform very different functions, they are closely connected. When the liver is not functioning properly, it can place significant stress on the heart and circulatory system of the body.
Liver disease not only affects the digestion process, metabolism, and detoxification but can also trigger serious cardiovascular changes that may go unnoticed in the early stages. Understanding this particular connection is significant for timely diagnosis and better overall care.
The Liver-Heart Connection
The liver helps to regulate the flow of blood, fluid balance, cholesterol metabolism, and inflammation as well. When liver disease develops—whether due to fatty liver, hepatitis, cirrhosis, or alcohol-related damage—all these processes become disturbed.
As a result of the same, the heart may have to work harder to maintain circulation, while blood vessels may also undergo certain changes that impact blood pressure and delivery of oxygen as well.
Changes In Blood Circulation
In well-advanced liver disease, especially cirrhosis, the flow of blood through the liver becomes restricted. This can also increase pressure in the portal vein, a condition known as portal hypertension. At the same point in time, blood vessels in the rest of the body may also widen, causing a drop in effective blood pressure.
To compensate, the heart pumps faster and harder. With the passage of time, this constant strain can weaken cardiac function and even lead to a condition sometimes referred to as cirrhotic cardiomyopathy, where the heart does not respond normally under stress.
Fluid Retention And Heart Stress
Liver disease often causes the body to retain both salt and water. This can also lead to swelling in the legs, abdomen, and surrounding tissues as well. Extra fluid in the body increases the workload on the heart, making it more difficult for it to pump blood in an efficient way. In severe cases, this may also contribute to shortness of breath, fatigue, and worsening cardiovascular strain.
Inflammation And Metabolic Impact
Some of the conditions, such as non-alcoholic fatty liver disease, are also linked with diabetes, obesity, high levels of cholesterol, and high blood pressure – all of which are the major risk factors for heart disease. Chronic inflammation, well-associated with liver damage, may further increase the risk of atherosclerosis and other cardiac complications.
GLP-1 Drugs: Why Not Everyone Taking Ozempic May Have Lower Blood Sugar Levels

Credit: iStock
GLP-1 receptor agonist drugs such as Ozempic and Wegovy have shown significant benefits for people with diabetes and obesity. However, not everyone experiences the same results.
Now, a study suggests that this variation may be due to genetic resistance, which means that some people are biologically less responsive to these medications, making these drugs less effective.
Also Read: NAFLD to MASLD: Experts Explain Why This Common Yet Dangerous Liver Condition Got Renamed
The new study by scientists at Stanford University in the US showed that about 10 per cent of the general population carry genetic variants that lead to what the researchers called “GLP-1 resistance”.
The GLP-1- receptor agonist drugs typically work by mimicking the hormone GLP-1 (glucagon-like peptide-1), which naturally helps regulate blood sugar by stimulating insulin release. The process slows the emptying of the stomach and reduces appetite.
However, in individuals with certain genetic variants, the team found that higher levels of GLP-1 did not regulate blood sugar better.
The researchers noted that the findings, published in the journal Genome Medicine, may be key to developing new therapies for people with GLP-1 resistance.
“There are a whole class of medications that are insulin sensitizers, so perhaps we can develop medications that will allow people to be sensitized to GLP-1s or find formulations of GLP-1, like the longer-acting versions, that avoid the GLP-1 resistance,” said Anna Gloyn, professor of pediatrics and of genetics at Stanford Medicine.
The Gene Linked to GLP-1 Resistance
Also read: Foundayo: US FDA Approves Eli Lilly’s GLP-1 Weight Loss Pill
To zero in on the gene resistant to GLP-1 drugs, the team conducted experiments in humans. They traced this resistance to a PAM variant known as p.S539W.
PAM (peptidyl-glycine alpha-amidating monooxygenase) is an enzyme that is uniquely capable of activating many hormones in the body, including GLP-1.
While the researchers suspected that people with the PAM variant would have lower levels of GLP-1 in their blood, it actually increased levels of GLP-1. Although the exact mechanism is still unclear, experiments in both humans and mice confirmed signs of reduced response to GLP-1.
Analyzing diabetes drug trial data, they found that individuals with these variants were less able to lower their blood sugar levels even after six months of treatment. This suggests that despite having more circulating GLP-1, their bodies are less responsive to it.
“When I treat patients in the diabetes clinic, I see a huge variation in response to these GLP-1-based medications and it is difficult to predict this response clinically,” said Mahesh Umapathysivam, an endocrinologist and clinical researcher at Adelaide University in Australia.
“This is the first step in being able to use someone’s genetic make-up to help us improve that decision-making process,” he added.
Also read: Can Weight Loss Jabs Surge Divorce Rates? What Experts Are Saying
Importantly, participants with the PAM variants did not respond differently to other common diabetes treatments, including sulfonylureas, metformin, and DPP-4i.
The finding may help develop precision medicine, the researchers said. Knowing ahead of time who is likely to respond would help patients get on the right drugs faster, Gloyn said.
Reasons Why Ozempic May Not Help All People Lose Weight
Ozempic is primarily indicated for type 2 diabetes management. But some doctors may prescribe it for weight loss in appropriate patients without diabetes.
Previous research has shown that medical conditions such as sleep apnea, along with certain common medications, such as antidepressants, steroids, and contraceptives, can hamper the process of shedding extra pounds.
Another major reason that can hamper weight loss is the side-effects of these drugs that can prompt a person to halt their prescription mid-way.
NAFLD to MASLD: Experts Explain Why This Common Yet Dangerous Liver Condition Got Renamed
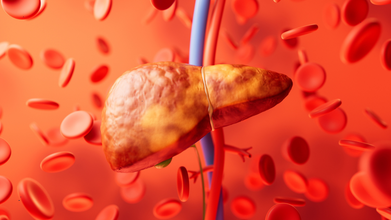
Credit: iStock
Once widely known as non-alcoholic fatty liver disease (NAFLD), the common and dangerous fatty liver condition was rephrased as Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) due to its strong link to metabolic health issues like obesity and diabetes.
MASLD now includes patients with fatty liver disease linked to metabolic risk factors such as obesity, diabetes, and hypertension.
Also Read: GLP-1 Drugs: Why Not Everyone Taking Ozempic May Have Lower Blood Sugar Levels
MASLD: So What Prompted The Change?
Globally, it was observed that all patients who have non-alcoholic fatty liver disease also have some associated form of metabolic dysfunction. The patients reported having either obesity, diabetes, metabolic syndrome, hypertension, or cholesterol problems.
And all these problems eventually lead to significant comorbidities later, like some people developed heart disease, while others developed complications of diabetes.
In view of these, a global consensus process in 2023 involving hundreds of experts from different countries adopted MASLD as it better reflected these underlying causes of the condition.
What Does MASLD Mean?
Also read: Lancet Study Shows Metabolic Liver Disease To Rise Over 38% By 2050: What’s Behind The Surge
MASLD is an umbrella term for liver conditions that develop in the presence of 1 or more cardiometabolic risk factors—including high blood sugar, elevated body mass index (BMI), and hypertension—but in the absence of other causes of liver fat accumulation.
The condition can be defined by excess liver fat accumulation (more than 5 per cent of liver weight) in the presence of metabolic dysfunction, independent of alcohol intake.
It encompasses a spectrum from simple steatosis to steatohepatitis, fibrosis, cirrhosis, and hepatocellular carcinoma.
“Initially, it was thought that having fatty liver disease without alcohol was a benign condition, but now it is recognized that, since it is associated with lots of metabolic comorbidities, it's no longer benign,” Dr. Ashish Kumar, Professor of gastroenterology and hepatology at Sir Ganga Ram Hospital in New Delhi, told HealthandMe.
He stated that whenever a diagnosis of fatty liver is present, "we should actually include other comorbidities, like obesity, dyslipidemia, which means cholesterol problem, diabetes, sugar problem, pre-diabetes, and hypertension. At least 50–70–80 percent of these patients will have one or more of these comorbidities".
Why Alcohol Is Not The Only Culprit For Fatty Liver
Although alcohol has remained the number one risk for liver disease, MASLD seems to be rising globally, including among people who do not drink. Why?
The reasons include:
a sedentary lifestyle,
increased consumption of fast and processed food,
lack of exercise,
lack of sleep,
stressful life.
Also read: Why Regular Scans Are Crucial for Liver Cancer Patients: Doctors Explain
The experts noted that food, especially the increasingly accessible junk food or processed food, is a major culprit.
“So even if the person is not drinking alcohol, people are developing addiction to processed food, and this is causing an epidemic level of obesity and diabetes. Consequently, MASLD is also increasing, and now it is becoming the number one cause of liver disease,” Dr Kumar said.
How To Prevent MASLD?
According to Dr. Sanjay Goja, Director, Liver Transplant & HPB Surgery, Narayana Hospital, Gurugram, prevention must focus on following a healthy lifestyle like maintaining a healthy BMI, engaging in regular physical activity, and eating a balanced diet.
Controlling diabetes, cholesterol, and blood pressure is also important to prevent the risk of MASLD.
Dr Siddharth Badola, Manipal Hospital, Ghaziabad, suggested sustainable lifestyle changes such as:
Maintaining an adequate body weight: Even slight weight loss (5–10 percent) has been shown to significantly reduce liver fat and inflammation.
Follow a balanced and nutrient-rich diet: People should focus on consuming whole grains, fresh fruits and vegetables, lean proteins, and healthy fats, while limiting refined carbohydrates and processed foods.
Avoid foods with added sugar: Excess consumption of fructose, commonly found in packaged foods and sugary beverages, is a key contributor to fat accumulation in the liver.
Engage in regular physical activity: At least 150 minutes of moderate-intensity exercise per week is recommended to improve insulin sensitivity and liver health.
Manage associated metabolic conditions: Effective control of diabetes, hypertension, and dyslipidemia is essential in reducing the risk of MASLD progression.
Ensure adequate sleep and stress management: Poor sleep quality and chronic stress can negatively impact metabolic balance and liver function.
Keep your body hydrated with ample water intake and follow structured meal timings.
© 2024 Bennett, Coleman & Company Limited

