- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
When Fear Clouds Judgment- The Psychology Behind The Mysterious Fog Over US And Canada

Image Credit: Canva (representational purpose only)
Mysterious Fog in the US , Canada and UK: A dense, eerie fog with a "burning chemical-like smell" has spooked a good part of North America and parts of the United Kingdom and Canada. With social media amplifying all concerns, this phenomenon has sparked attention across all social media platforms. However, at the heart of this mysterious fog are a conjunction of natural events, social psychology, and environmental conditions that culminated in all the conspiracy theories and public health fears. Here's a closer look at the mysterious fog, its potential causes, and the societal response it has triggered.
Fog that Feeds Fear
The first reports of this "mysterious fog" came in from Florida where a resident said that they experienced respiratory symptoms, feverish warmth, and stomach cramps after contact with the fog. Similar stories started flooding social media, and within a day or two, a sinister force seemed to sweep across the United States, Canada, and parts of the UK. From Texas to Minnesota, people reported weird odors and health issues that they thought were linked to this bizarre atmospheric event.
Some witnesses were said to see "white particles" swirling through the air; theories ranged from a chemical attack or experimental weapon to drone-related chemical dispersals and references to historical military experiments, such as the infamous 1950s "Operation Sea-Spray."
Fuel to the fire were added when videos and posts, hundreds of thousands in number, began circulating on social media sites like TikTok and X (formerly Twitter) speculating on the origin of the fog. Hashtags like #ToxicFog went trending for days. Hysteria created a self-reinforcing loop in which every post spurred further scrutiny and fear.
Scientific Explanation of the Dense Mysterious Fog
1. What is Fog?
Fog is essentially a low-lying cloud formed when the air temperature cools to its dew point, causing water vapor to condense into tiny droplets or ice crystals. Several types of fog—advection fog, radiation fog, and valley fog—can form depending on conditions such as warm, moist air moving over cooler land or when temperatures plummet rapidly under clear skies.
2. Why the Chemical Smell?
Such chemical-like smell as reported during the occurrence of fog events is sometimes attributed to air pollution. It acts like a sponge, where it absorbs these pollutants, which include sulfur dioxide and nitrogen oxides, among others, that emit from industries. This mixture, therefore, leads to a stench that could be mistaken as unnatural or even toxic.
Also Read: Health Concerns Rise As US, Canada, and UK Come Under The Blanket Of Thick, Dense, Toxic Fog
3. What are the Health Concerns?
High moisture levels from fog can significantly exacerbate symptoms related to respiration, but especially in already predisposed asthmatics and allergy patients. The connection of these symptoms with actual fever, stomach cramps, and puffy eyes is too remote. Experts assume that the irritating effects of entrapped pollutants trapped in fog tend to affect more the eyes and throat rather than the rest of the body affected by some report.
Psychological Effects of Mass Panic Caused by Social Media
Social media amplified a natural weather event into a health epidemic. It made the personal experience of individuals become a cause for fear and speculation, a domino effect.
According to psychologists, this is a concept of selective perception, wherein once people's attention is drawn to environmental anomalies, they begin to notice them. This mirrors earlier panics, such as the Seattle windshield pitting panic of 1954. Then, atomic bomb testing caused fear in many and started to have people looking at their windshields for small marks that they had not seen before. Likewise, postings on the strangeness of the fog probably increased public awareness and suspicion, with people looking to attach unrelated symptoms to the phenomenon.
The fog hysteria shares a commonality with other instances of mass panic, such as the "drone sightings" of recent years or the Cold War-era fears of biological warfare.
Also Read: Mysterious Fog Is Making Americans Sick
These events underscore how fear can cloud judgment, especially when amplified by social media and sensationalist headlines. While historical cases, such as "Operation Sea-Spray," offer concrete evidence of the existence of unethical experiments, the jump from a natural weather condition to theories of chemical attacks exemplifies a more modern trend of connecting unrelated dots, all wonderfully seeded in distrust and anxiety.
Despite the swirling rumors, meteorologists and scientists are in agreement that the mysterious fog is not as alarming as it seems. It is well known that fog traps and amplifies pollutants, especially in urban and industrial regions. Moreover, winter months are the most conducive for fog formation, so its recent prevalence is unsurprising.
On the other hand, environmentalists advise that the fog should wake everyone up to increased levels of pollution. The reported odors and health irritations could be just symptoms of far deeper systemic issues like industrial emissions and lack of control over air quality.
The authorities must be transparent in their communication to combat misinformation and allay public fears. Governments and environmental agencies must provide timely updates on weather phenomena, air quality, and health risks. Initiatives like real-time pollutant tracking and public education campaigns can help demystify natural occurrences while addressing valid environmental concerns.
The mysterious Canada fog is a compelling case study in how environmental events intersect with psychology and societal dynamics. While rooted in natural phenomena, the fog became a vessel for collective fears, amplified by modern technology and historical anxieties.
In this information era where communication occurs at an almost lightning pace, the fog becomes a metaphor that reminds everyone about scientific literacy, environmental responsibility, and an effective balance when considering public concern. Whether perceived as a marvel of nature or as a tale that serves to teach, it left a very powerful mark in people's minds.
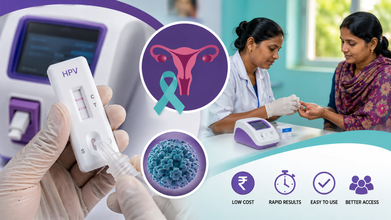
Affordable India-Made HPV Test Offers Hope For Early Cervical Cancer Detection
Credit: AI generated image
An indigenous low-cost point-of-care (PoC) testing approach has shown promise in making screening for Human Papillomavirus (HPV) more affordable and accessible, potentially improving the early detection of cervical cancer, according to a joint study led by researchers from AIIMS, ICMR, and the WHO's International Agency for Research on Cancer (IARC).
The study, published in the International Journal of Cancer, found that PoC tests have the potential to become scalable, low-cost screening tools in India and other low- and middle-income countries (LMICs).
The objective of the study was to evaluate indigenous HPV DNA testing platforms for use in primary cervical cancer screening.
Four Indigenous Tests Evaluated
While several Indian HPV tests have been developed, many had not yet been validated against the international standards laid down by the WHO.
The study evaluated four point-of-care tests developed by 3 Indian companies:
- Genes2Me – HPV-Q
- Mylabs India – PathoDetect HPV-7 and PathoDetect HPV-14
- Molbio Diagnostics – Truenat HR-HPV-Plus
Each test was developed by repurposing existing COVID-19 RT-PCR platforms. HPV-Q and PathoDetect HPV-14 can detect all 14 high-risk HPV types, while PathoDetect HPV-7 detects seven high-risk types: 16, 18, 31, 33, 45, 52, and 58.
How the Tests Performed
Also read: PM Modi Launches Nationwide Free HPV Vaccination Drive; A Landmark Step, Says WHO
To assess performance, researchers analyzed 1,159 cervical samples collected from studies conducted in Argentina and Costa Rica.
The results showed:
- Truenat HR-HPV-Plus: Sensitivity 80.4%, specificity 91.5% — met IARC validation criteria.
- PathoDetect HPV-7: Sensitivity 68.1%, specificity 89.0% — lower sensitivity.
- HPV-Q and PathoDetect HPV-14: Did not meet validation criteria.
Indigenous Platforms Can Be Game-Changers
"This study demonstrates that indigenous platforms can achieve clinical validation, quality standards, and accuracy comparable to global gold-standard tests. It will be a game-changer for cervical cancer prevention and early detection in India," said lead author Dr Neerja Bhatla, Professor Emeritus, NCI and former Head of the Department of Obstetrics & Gynecology at AIIMS New Delhi.
Dr Shalini Singh, Director of ICMR-NICPR, added that HPV DNA testing at age 35 and again at age 45 is central to WHO's cervical cancer elimination strategy, but high costs remain a major barrier in LMICs.
"Affordable indigenous solutions are essential to expand screening coverage and reduce disease burden," she said.
High Burden Of Cervical Cancer: Why HPV Testing Matters
Read More: HPV Vaccine Can Help Curb Rising Head And Neck Cancers, Says Top US Doctor
Cervical cancer continues to be a major public health challenge in India, with around 127,000 new cases and 80,000 deaths reported annually.
Globally, cervical cancer is the fourth most common cancer in women. According to WHO data for 2022, there were approximately 662,000 new cases and 348,000 deaths worldwide.
Regular screening of women over 30 years of age every 3-5 years can help detect precancerous and cancerous lesions early. However, screening coverage in India has remained low despite the inclusion of visual inspection with acetic acid (VIA) in the national screening program for breast, oral, and cervical cancers.
Persistent infection with carcinogenic HPV types is the necessary cause of cervical cancer. WHO recommends a transition to HPV testing as the preferred strategy for cervical cancer elimination.
Using validated HPV tests, only two rounds of screening — at ages 35 and 45 — may be sufficient to help achieve elimination targets. However, most existing HPV tests are expensive, require sophisticated laboratory infrastructure, and are not easily available at last-mile health facilities.
The researchers said that the point-of-care HPV testing approach, thus, could be a major breakthrough, enabling affordable diagnostics and even same-day treatment for women who test positive.
Potential for Nationwide Rollout
The researchers noted that while the recently introduced national HPV vaccination campaign will benefit younger generations, there remains an urgent need to expand HPV testing for women over 30 who are currently at risk of cervical cancer.
Dr Showket Hussain of the ICMR-National Institute of Cancer Prevention and Research (NICPR), Noida, said the validated platform has strong potential for integration into national screening programs because district-level healthcare personnel are already familiar with similar testing systems.
"Future advancements such as self-sampling and high-throughput adaptations could further enhance accessibility," he said.
The findings are expected to support India's cervical cancer elimination goals while offering a scalable model for other low-resource settings globally.
Cancer Patients To Face Higher Costs As India Raises Chemotherapy Drug Prices Amid Nationwide Shortage
Credit: iStock
Cancer patients in India, already affected by shortages of critical chemotherapy drugs, are now set to face higher costs after the government approved a price hike.
Facing significant shortages are Cisplatin and Carboplatin — the two platinum-based drugs that form the backbone of treatment for common cancers such as oral cancer, breast cancer, ovarian cancer, cervical cancer, esophageal cancer, and testicular cancer.
Shortages Disrupt Cancer Treatment
The shortage of these essential chemotherapy medicines is disrupting care across hospitals and cancer centers, with several doctors pointing out that a substantial proportion of chemotherapy protocols for solid tumors rely on one of these platinum-based agents.
The drugs have been facing shortages as companies find it difficult to sustain production amid rising manufacturing costs linked to the ongoing conflict in the Middle East.
The two drugs, often prescribed in combination and with no alternatives, are derived from platinum, a precious metal whose cost has surged due to the Middle East conflict and the closure of the Strait of Hormuz.
Rising Production Costs Hit Manufacturers
Also read: India Revises Folic Acid Measurement Unit to Prevent Dosage Errors
"The production cost has increased because the API, or active pharmaceutical ingredient, which is imported from abroad, has become more expensive. As a result, manufacturing costs have gone up, but the MRP has not increased," Dr Shyam Agarwal, Senior Consultant, Medical Oncology at Sir Ganga Ram Hospital, told HealthandMe.
As companies have struggled to sustain production amid rising manufacturing costs, they approached the government seeking a revision of the price cap.
Drugmakers reportedly sought price revisions for 82 medicines, citing substantial increases in production costs due to the US-Iran conflict linked to the Hormuz crisis and other factors.
Government Approves Higher Prices
In response, the National Pharmaceutical Pricing Authority (NPPA), following permission from the Union government's Department of Pharmaceuticals (DoP), has increased maximum retail prices for four drugs, including the life-saving cancer medicines Cisplatin and Carboplatin, and two anti-tetanus injections.
While the move is aimed at restoring supplies, it has also raised concerns about affordability for patients already battling a costly disease.
The NPPA is the country's drug price regulator and functions under the DoP, Ministry of Chemicals and Fertilizers.
NPPA Asked to Review Cost Increases
Read More: India's Maternal Mortality Ratio Drops From 130 To 87 In Last 10 Years: Govt
The ministry directed the pricing authority to determine how much prices could be increased.
The formula suggested: "The Standing Committee recommended a 10 per cent increase per year from the last fixation with a ceiling of 50 per cent, which could also be a guide, but the primary principle should be cost increase," News18 reported.
The DoP has further advised the NPPA to examine increases in raw material costs for these drugs while determining any price revisions. It has also asked the regulator to assess similar requests from the pharmaceutical industry, where price hikes have been sought due to higher raw material costs.
India Revises Folic Acid Measurement Unit to Prevent Dosage Errors

Credit: iStock
India's Ministry of Health has notified an amendment to the Drugs Rules, 1945, replacing the unit of measurement for folic acid under Schedule V from "mg" (milligrams) to "mcg" (micrograms).
The amendment has been brought into effect through the Drugs (Fourth Amendment) Rules, 2026, notified vide Gazette Notification G.S.R. 444(E) dated June 1, 2026. The notification will come into force after six months, according to the Ministry.
The move aims to avoid unnecessary confusion, reduce the chances of misinterpretation, and prevent incorrect administration of dosages of vitamin B9 — essential for the production of healthy red blood cells, especially during pregnancy.
The change follows the publication of draft rules in January 2026 and consideration of objections and suggestions received from stakeholders.
What Does The New Notification Say?
Also read: India Makes Significant Progress In Maternal Care, Nutrition And Child Immunization: NFHS-6
As per the notification, in Schedule V of the Drugs Rules, 1945, under paragraph 2 relating to standards for patent or proprietary medicines, the entry against "Folic acid" in the unit column has been amended. The earlier unit "mg" has now been substituted with "mcg".
The amendment will correct the unit of measurement, bringing it in line with internationally accepted dosing standards for folic acid, which is routinely prescribed and labelled in micrograms. This correction is also important because lower doses of folic acid are used prophylactically in situations such as pregnancy to prevent infant malformations. Higher doses, however, may be used to address specific conditions such as neural tube defects, Medical Dialogues reported.
The Ministry stated that the amendment has been made after consultation with the Drugs Technical Advisory Board (DTAB) and in exercise of the powers conferred under Sections 12 and 33 of the Drugs and Cosmetics Act, 1940.
According to the notification, the Drugs (Fourth Amendment) Rules, 2026, will come into force six months from the date of publication in the Official Gazette, giving manufacturers and other stakeholders sufficient time to make necessary compliance-related changes in product documentation, labelling, and regulatory records.
Why Is Folic Acid Important?
Read More: Introducing Eggs Before Age One May Lower Allergy Risk by 17%: Study
According to the American College of Obstetricians and Gynecologists (ACOG), folic acid is one of the most important nutrients in prenatal vitamins. This B vitamin helps create the baby's neural tube — the structure that eventually forms the brain and spinal cord.
Beyond pregnancy, people who do not consume enough folate- or folic acid-rich foods may develop folate deficiency. People with celiac disease and inflammatory bowel disease, which prevent the small intestine from absorbing nutrients properly, may also have low folate levels, as per the Mayo Clinic.
Folate is found mainly in
- dark green leafy vegetables,
- beans,
- peas,
- nuts.
Fruits rich in folate include
- oranges,
- bananas,
- melons,
- papayas.
The recommended daily amount of folate for adults is 400 micrograms (mcg). People who are planning a pregnancy or could become pregnant should get 400 to 800 mcg of folic acid daily.
Importantly, taking too much folic acid, especially over 1,000 mcg daily without a doctor's advice, can mask a vitamin B12 deficiency, potentially causing irreversible nerve damage and may lead to side effects such as bitter taste, nausea or sleep problems, with potential links to increased risks in pregnancy like autism or insulin resistance.
© 2024 Bennett, Coleman & Company Limited

