- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Matthew Perry Investigation: Can Ketamine Kill Someone?

Credits: IMDb
“I'm not great at the advice. Can I interest you in a sarcastic comment?”

This is what ‘Friends’ actor Matthew Perry’s character Chandler Bing was known for. He was known for being funny. However, he had his own struggles in his personal life and those struggles were acute depression. He was treating it with ketamine infusion therapy which is legal in the US and the UK.
What is Ketamine infusion therapy?
Ketamine is an anaesthetic used to treat depression, anxiety and pain under supervised and controlled medical settings. However, it does have its side effects, which can lead to distortion of sight, sound and time. It can also produce calming and relaxing effects.
Ketamine increases a person’s heart rate and blood pressure. If overdosed, it can leave users confused and agitated and can cause them to hurt themselves without even realising it. It can also lead to liver damage and bladder problems.
However, when used in moderation and under the supervision of medical doctors, it can treat depression where traditional antidepressants have failed.
Prof Rupert McShane, a University of Oxford psychiatrist who runs an NHS ketamine treatment clinic told BBC that ketamine “probably turns off the area of the brain that is involved in disappointment.”
Can Ketamine Infusion Therapy Kill Someone?
In simple terms, it cannot, be if the dosage is given in a controlled setting and as prescribed. Ketamine infusion therapy uses drugs in small doses than those used for anaesthesia. It acts faster than traditional anti-depressants, but the effects also wear off way quickly. Which is why it is important to monitor patients’ mental state for relapsing back into depression and discouraging them from overdosing on it.
There are ways of giving people ketamine. One of the ways is through “infusing”, which means to use an IV drip. However, injections, nasal sprays and capsules are also methods used to give people ketamine.
Since the dosage of ketamine used in the infusion treatment is small, it being the reason of actor Perry’s death was ruled out. The medical examiner also noted that Perry’s last ketamine infusion therapy session happened more than a week before his death, which means by the time he had died, it must have worn off.
So, What Happened To Perry?
Though Perry’s last session was more than a week before, his post-mortem showed that his blood contained a high concentration of ketamine. He had died of the “acute effects” of ketamine.
If it was not his session, then how did he get ketamine?
Prosecutors alleged that his assistant gave him at least 27 shots of ketamine in four days before his death, reported BBC.
Perry has been open about his personal struggles and this is what the doctors and dealers used against him. Martin Estrada, the US attorney for California’s Central District told the BBC that people took advantage of his condition. They charged him 165 times more than what vials of ketamine cost.
Names that have come up include Dr Salvador Plasencia, drug dealers “Ketamine Queen” aka Jasveen Sangha and Eric Fleming, and Perry’s live-in assistant Kenneth Iwamasa.
Who Are These Names And What Did They Do?
Ketamine Queen or Sangha supplied drugs that led to Perry’s death. Her home was a “drug-selling emporium,” said Estrada. More than 80 vials of ketamine, and thousands of pills including methamphetamine, cocaine and Xanax were allegedly found in her house known as the “Sangha Stash House.”
Sangha is known to deal with high-end celebs and was a “major source of supply for ketamine to others as well as Perry,” said Estrada.
Dr Plasencia called Perry a “moron” while charging him $2,000 for vials that cost only $12. He sold Perry 20 vials of ketamine between September and October 2023, costing $55,000.
He was the one who taught Iwamasa, who had no medical knowledge to inject the drug. This is after he knew that “Perry’s ketamine addiction was spiralling out of control,” as per what the investigators told the BBC.
Another dealer Fleming was told by Sangha to “delete all our messages.” While Fleming pleaded guilty to conspiring to distribute drugs unlawfully, he also allegedly messaged Sangha: “Please call...Got more info and want to bounce ideas off you. I’m 90% sure everyone is protected. I never dealt with [Perry] only his assistant. So the assistant was the enabler.”
The court documents also revealed that he asked Sangha on whether the ketamine stays in your system or “is it immediately flushed out.”
Dr Pepper, Bots, Cans
The people who allegedly exploited Perry used coded language for ketamine and called it “Dr Pepper”, “bots”, or “cans.”
Selling overpriced drugs, taking advantage of Perry’s mental condition and falsifying medical records to make the drugs given to him look legitimate by Dr Plasencia is what took Perry’s life.
Iwamasa is said to have administered more than 20 shots of ketamine and three on the day Perry died. Whereas ketamine is only administered by a physician. Authorities also found that weeks before Perry’s death, Dr Plasencia allegedly bought 10 vials of ketamine and intended to sell to Perry.
He also injected Perry with a large dose, two days later. This caused him to “freeze up” and spiked his blood pressure.
When I Die, I Want Helping Others To Be The First Thing That’s Mentioned
Perry had always been open about his drug addictions, struggles with alcohol and his depression. He said that his openness would help others who are also struggling and wanted to be remembered by his quote which also is on the homepage of the Mattew Perry Foundation that helps others struggling with the disease of addiction: “When I die, I want helping others to be the first thing that’s mentioned.”
Five arrests have been made in the case so far.
X-Men Star Tyler Mane Reveals ‘Super-Rare’ Male Breast Cancer Diagnosis

Credit: Tyler Mane/ Instagram
X-Men actor Tyler Mane has revealed that he has been diagnosed with a "super-rare" form of breast cancer.
In a post on Instagram, Mane, a former professional wrestler, shared that he has been diagnosed with the condition, which is predominantly associated with women. He noted that only about 1 per cent of breast cancer cases occur in men.
The 59-year-old actor said he initially considered keeping his diagnosis private, but ultimately decided to speak publicly about it because men are often diagnosed at more advanced stages of the disease due to a lack of awareness.
“I have some bad news: I start chemo today. One in 750 men will be diagnosed with breast cancer in their lifetime and I’m one of them,” he said in a video posted on Instagram.
Mane revealed that doctors initially dismissed the lump he discovered.
“My doctors initially dismissed it and it was only because my wife pushed me to get the lump removed that I got in early,” he said.
The actor admitted that his first instinct was to keep the diagnosis secret.
“I’ll be honest, my first reaction was to keep it secret. I mean it’s kind of embarrassing,” he said.
However, after learning more about male breast cancer and the importance of early detection, he decided to share his experience publicly to help raise awareness.
While Mane did not reveal the stage of his cancer, he informed of undergoing chemotherapy and urged his followers to spread awareness about the disease.
“I’m gonna kick cancer’s ass. Thank you for coming along for the journey. We need to spread the awareness. Cancer sucks but if you catch it quick enough, you can win this battle,” he wrote.
Breast Cancer in Men
According to the CDC, about one in every 100 breast cancer cases diagnosed in the United States occurs in men. The situation is similar globally, with the World Health Organization estimating that between 0.5 and 1 per cent of breast cancers occur in men.
The lifetime risk of breast cancer in men is currently less than one in 1,000. However, the incidence of the disease is increasing, particularly with advancing age.
The average age at diagnosis for men is about 67 years, roughly five years later than the average age of diagnosis in women.
Because breast cancer is often not suspected in men, the condition can be overlooked or misdiagnosed, resulting in many cases being detected at later stages.
What Are the Risk Factors in Men?
Several factors can increase a man's likelihood of developing breast cancer. These include:
- Advancing age
- Genetics
- Family history
- Lifestyle and hormonal factors, including excess weight, alcohol consumption, liver disease and hormonal imbalances
- Medical conditions such as gynecomastia and Klinefelter syndrome
- Radiation exposure, particularly previous radiation treatment to the chest area
Symptoms to Watch For
The symptoms of breast cancer in men are similar to those in women, but men may overlook them because they are less aware of the disease.
Common signs include:
- A lump in the breast, usually painless
- Nipple discharge
- Dimpling, redness or inversion of the skin or nipple
Why Early Detection Matters
Routine mammography is not generally recommended for men because of the low prevalence of the disease. However, men considered to be at high risk, including those with a strong family history or genetic predisposition, may benefit from periodic screening and should discuss appropriate options with their healthcare providers.
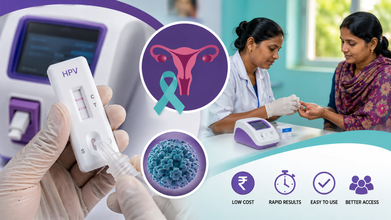
Affordable India-Made HPV Test Offers Hope For Early Cervical Cancer Detection
Credit: AI generated image
An indigenous low-cost point-of-care (PoC) testing approach has shown promise in making screening for Human Papillomavirus (HPV) more affordable and accessible, potentially improving the early detection of cervical cancer, according to a joint study led by researchers from AIIMS, ICMR, and the WHO's International Agency for Research on Cancer (IARC).
The study, published in the International Journal of Cancer, found that PoC tests have the potential to become scalable, low-cost screening tools in India and other low- and middle-income countries (LMICs).
The objective of the study was to evaluate indigenous HPV DNA testing platforms for use in primary cervical cancer screening.
Four Indigenous Tests Evaluated
While several Indian HPV tests have been developed, many had not yet been validated against the international standards laid down by the WHO.
The study evaluated four point-of-care tests developed by 3 Indian companies:
- Genes2Me – HPV-Q
- Mylabs India – PathoDetect HPV-7 and PathoDetect HPV-14
- Molbio Diagnostics – Truenat HR-HPV-Plus
Each test was developed by repurposing existing COVID-19 RT-PCR platforms. HPV-Q and PathoDetect HPV-14 can detect all 14 high-risk HPV types, while PathoDetect HPV-7 detects seven high-risk types: 16, 18, 31, 33, 45, 52, and 58.
How the Tests Performed
Also read: PM Modi Launches Nationwide Free HPV Vaccination Drive; A Landmark Step, Says WHO
To assess performance, researchers analyzed 1,159 cervical samples collected from studies conducted in Argentina and Costa Rica.
The results showed:
- Truenat HR-HPV-Plus: Sensitivity 80.4%, specificity 91.5% — met IARC validation criteria.
- PathoDetect HPV-7: Sensitivity 68.1%, specificity 89.0% — lower sensitivity.
- HPV-Q and PathoDetect HPV-14: Did not meet validation criteria.
Indigenous Platforms Can Be Game-Changers
"This study demonstrates that indigenous platforms can achieve clinical validation, quality standards, and accuracy comparable to global gold-standard tests. It will be a game-changer for cervical cancer prevention and early detection in India," said lead author Dr Neerja Bhatla, Professor Emeritus, NCI and former Head of the Department of Obstetrics & Gynecology at AIIMS New Delhi.
Dr Shalini Singh, Director of ICMR-NICPR, added that HPV DNA testing at age 35 and again at age 45 is central to WHO's cervical cancer elimination strategy, but high costs remain a major barrier in LMICs.
"Affordable indigenous solutions are essential to expand screening coverage and reduce disease burden," she said.
High Burden Of Cervical Cancer: Why HPV Testing Matters
Read More: HPV Vaccine Can Help Curb Rising Head And Neck Cancers, Says Top US Doctor
Cervical cancer continues to be a major public health challenge in India, with around 127,000 new cases and 80,000 deaths reported annually.
Globally, cervical cancer is the fourth most common cancer in women. According to WHO data for 2022, there were approximately 662,000 new cases and 348,000 deaths worldwide.
Regular screening of women over 30 years of age every 3-5 years can help detect precancerous and cancerous lesions early. However, screening coverage in India has remained low despite the inclusion of visual inspection with acetic acid (VIA) in the national screening program for breast, oral, and cervical cancers.
Persistent infection with carcinogenic HPV types is the necessary cause of cervical cancer. WHO recommends a transition to HPV testing as the preferred strategy for cervical cancer elimination.
Using validated HPV tests, only two rounds of screening — at ages 35 and 45 — may be sufficient to help achieve elimination targets. However, most existing HPV tests are expensive, require sophisticated laboratory infrastructure, and are not easily available at last-mile health facilities.
The researchers said that the point-of-care HPV testing approach, thus, could be a major breakthrough, enabling affordable diagnostics and even same-day treatment for women who test positive.
Potential for Nationwide Rollout
The researchers noted that while the recently introduced national HPV vaccination campaign will benefit younger generations, there remains an urgent need to expand HPV testing for women over 30 who are currently at risk of cervical cancer.
Dr Showket Hussain of the ICMR-National Institute of Cancer Prevention and Research (NICPR), Noida, said the validated platform has strong potential for integration into national screening programs because district-level healthcare personnel are already familiar with similar testing systems.
"Future advancements such as self-sampling and high-throughput adaptations could further enhance accessibility," he said.
The findings are expected to support India's cervical cancer elimination goals while offering a scalable model for other low-resource settings globally.
Cancer Patients To Face Higher Costs As India Raises Chemotherapy Drug Prices Amid Nationwide Shortage
Credit: iStock
Cancer patients in India, already affected by shortages of critical chemotherapy drugs, are now set to face higher costs after the government approved a price hike.
Facing significant shortages are Cisplatin and Carboplatin — the two platinum-based drugs that form the backbone of treatment for common cancers such as oral cancer, breast cancer, ovarian cancer, cervical cancer, esophageal cancer, and testicular cancer.
Shortages Disrupt Cancer Treatment
The shortage of these essential chemotherapy medicines is disrupting care across hospitals and cancer centers, with several doctors pointing out that a substantial proportion of chemotherapy protocols for solid tumors rely on one of these platinum-based agents.
The drugs have been facing shortages as companies find it difficult to sustain production amid rising manufacturing costs linked to the ongoing conflict in the Middle East.
The two drugs, often prescribed in combination and with no alternatives, are derived from platinum, a precious metal whose cost has surged due to the Middle East conflict and the closure of the Strait of Hormuz.
Rising Production Costs Hit Manufacturers
Also read: India Revises Folic Acid Measurement Unit to Prevent Dosage Errors
"The production cost has increased because the API, or active pharmaceutical ingredient, which is imported from abroad, has become more expensive. As a result, manufacturing costs have gone up, but the MRP has not increased," Dr Shyam Agarwal, Senior Consultant, Medical Oncology at Sir Ganga Ram Hospital, told HealthandMe.
As companies have struggled to sustain production amid rising manufacturing costs, they approached the government seeking a revision of the price cap.
Drugmakers reportedly sought price revisions for 82 medicines, citing substantial increases in production costs due to the US-Iran conflict linked to the Hormuz crisis and other factors.
Government Approves Higher Prices
In response, the National Pharmaceutical Pricing Authority (NPPA), following permission from the Union government's Department of Pharmaceuticals (DoP), has increased maximum retail prices for four drugs, including the life-saving cancer medicines Cisplatin and Carboplatin, and two anti-tetanus injections.
While the move is aimed at restoring supplies, it has also raised concerns about affordability for patients already battling a costly disease.
The NPPA is the country's drug price regulator and functions under the DoP, Ministry of Chemicals and Fertilizers.
NPPA Asked to Review Cost Increases
Read More: India's Maternal Mortality Ratio Drops From 130 To 87 In Last 10 Years: Govt
The ministry directed the pricing authority to determine how much prices could be increased.
The formula suggested: "The Standing Committee recommended a 10 per cent increase per year from the last fixation with a ceiling of 50 per cent, which could also be a guide, but the primary principle should be cost increase," News18 reported.
The DoP has further advised the NPPA to examine increases in raw material costs for these drugs while determining any price revisions. It has also asked the regulator to assess similar requests from the pharmaceutical industry, where price hikes have been sought due to higher raw material costs.
© 2024 Bennett, Coleman & Company Limited

