- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Centre Has Banned 156 Medicines, Including Paracetamol, Due To Increasing Health Risk

Banned medicine (Credit-Canva)
The Union Health Ministry implemented a ban on 156 "irrational" FDC medicines, effective immediately. These medicines, including widely used antibiotics, painkillers, and multivitamins, were commonly used to treat fever, cough, and infections. The ban was imposed due to the associated health risks and lack of therapeutic justification for the ingredients in these FDCs.
What are FDC medicines?
FDCs or fixed-dosed combinations, also known as "cocktail drugs," are medications that combine multiple drugs in a single pill. They are designed to treat multiple symptoms or conditions simultaneously. While they offer convenience, they can pose significant risks. These risks include the possibility of overdose, adverse interactions between the drugs, and the development of antibiotic resistance. Additionally, many FDC medicines lack sufficient scientific evidence to support their safety and efficacy.
Expert opinions on FDC medicines
Experts have raised concerns about the use of FDC medicines. They believe that many of these combinations lack sufficient scientific evidence to support their safety and effectiveness. Additionally, the combination of multiple drugs in a single pill can increase the risk of adverse side effects and interactions with other medications.
Experts have also found that FDC medicines may not be as effective as individual drugs in treating certain conditions. It is important to note that safer and more effective alternatives are available for most of the medical conditions that FDC medicines were used to treat. One particular concern is the inclusion of antibiotics in some FDCs. Overuse of antibiotics can contribute to the development of antibiotic resistance, a major public health threat.
The risks of FDC medicines
The use of FDC medicines can lead to adverse effects, including serious ones. Additionally, safer alternatives, tested in clinical trials, are available to treat the same medical conditions. Experts recommend prescribing drugs individually based on a patient's clinical symptoms rather than combining them in FDCs.
The ban on irrational FDC medicines by the Union Health Ministry can be seen as a positive step towards promoting rational drug use and protecting public health. The goal is to eliminate unnecessary and potentially harmful drug combinations. This is a step forward in reducing the risks associated with medication and ensure safer and more effective treatment options for patients.
Kerala Records 85 Shigella Cases This Year: Know The Symptoms And Risks
Credit: Canva
Kerala is battling a highly contagious bacterial disease called shigellosis (bacillary dysentery), caused by the bacteria Shigella. The southern state has recorded 85 cases of the intestinal infection called shigellosis since the beginning of this year.
All districts in the state have reported sporadic cases of the disease, which is primarily caused by contaminated food and water or poor hygiene practices.
Currently, cases have been reported from northern districts such as Wayanad, Kozhikode, Alappuzha, and Malappuram.
This week, two cases involving a four-and-a-half-year-old boy and a five-year-old girl were confirmed among children at Mar Baselios School in Wayanad.
About 25 students with suspected Shigella infection have been admitted to the Sultan Bathery Taluk Headquarters Hospital. However, Kerala Health Minister K. Muraleedharan said their conditions are stable, ANI reported.
Earlier, a four-year-old girl undergoing treatment at Kozhikode Medical College died after being infected with Shigella.
Also read: Kerala Reports 5 Suspected West Nile Fever Cases: Know All About The Mosquito-borne Disease
According to health authorities, 339 people have reported symptoms associated with the infection. Of these, 21 are undergoing treatment at Sultan Bathery Taluk Headquarters Hospital, while 38 others are receiving treatment at private hospitals.
Officials said none of the patients is currently in serious condition. A total of 21 samples were sent for laboratory testing. Two samples have tested positive so far, while the results of the remaining samples are awaited.
Muraleedharan also asserted that the Shigella outbreak reported in parts of the state remains under control, even as health authorities continue monitoring cases in Wayanad and other districts.
The Health Minister noted that a special medical team from Kozhikode Medical College has been deployed to Wayanad, where several students were admitted to hospital following reports of shigellosis, a highly contagious bacterial intestinal infection.
Shigella: Why Are Young Children at High Risk?
Speaking to HealthandMe, Dr. Rajeev Jayadevan, Ex-President of IMA Cochin and Convener of the Research Cell, Kerala, explained that even a small number of bacteria—as few as 10—is enough to cause infection.
It is because the Shigella bacteria “is able to withstand the hostile acidic environment of the human stomach. Outside the human body, it can stay alive in water for weeks together,” he said.
Shigella is most severe in children below the age of five, as their immune systems are less efficient at clearing the infection early.
“Adults also become ill, but acquired immunity from childhood keeps the infection mild in many instances,” the expert said.
How Does Shigella Spread?
Read More: Confused By Your Cholesterol Report? Here's What LDL And ApoB Really Mean
Shigella spreads through:
- Touching contaminated surfaces or objects
- Physical contact with an infected individual
- Drinking contaminated water or ice made from unclean water
“Shigella outbreaks can occur in crowded places such as anganwadis, where children come into close physical contact with each other and share toys. Very young children also have the habit of putting their fingers in their mouths, which enables the bacteria to gain access to their gut,” Dr. Jayadevan said.
The expert noted that houseflies can also spread the bacteria. Flies transmit the bacteria from human feces to food, water, and surfaces, contributing to up to 37 percent of cases in some regions, according to a study published in PLOS Neglected Tropical Diseases.
Shigella: What Are the Symptoms?
The disease typically develops within one to three days after ingestion of the bacteria and may begin with:
- Fever
- Diarrhea
- Body aches
- Headache
Once the infection involves the large intestine, bloody diarrhea can occur along with severe abdominal cramps.
How to Prevent Shigella
Dr. Jayadevan stressed the need for early recognition and treatment with appropriate antibiotics. ORS can help prevent dehydration, while zinc tablets can enhance recovery.
Other measures to reduce the risk of spread include:
- Washing hands with soap and water, especially before touching food and after using the toilet
- Maintaining hygiene among food handlers
- Controlling flies.
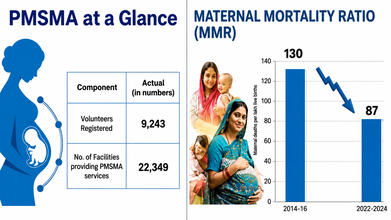
India's Maternal Mortality Ratio Drops From 130 To 87 In Last 10 Years: Govt
Credit: Health Ministry/PIB
From 130 maternal deaths per lakh live births during 2014–16, India has achieved a 43-point reduction in the Maternal Mortality Ratio (MMR) between 2022 and 2024, according to the government.
Currently, the MMR in the country stands at 87 maternal deaths per lakh live births.
In an official statement, the government credited the achievement to Pradhan Mantri Surakshit Matritva Abhiyaan (PMSMA), which today completed a decade.
Launched in June 2016, PMSMA provides free, comprehensive antenatal care to pregnant women at designated government health facilities on the 9th of every month.
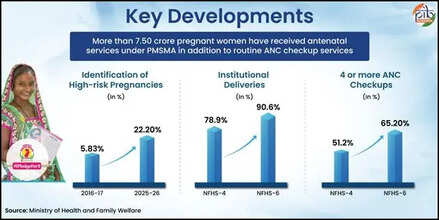
More than 7.50 crore pregnant women have received antenatal services under PMSMA in addition to routine ANC check-up services in the last 10 years. The Extended PMSMA, launched in 2022, strengthens follow-up care and tracking for high-risk pregnancies.
"Over the past decade, the Pradhan Mantri Surakshit Matritva Abhiyaan has transformed the delivery of antenatal care in India. Quality maternal healthcare is now more accessible, systematic and responsive," the statement said.
“The remarkable decline in the Maternal Mortality Ratio from 130 to 87 over the past decade demonstrates how focused interventions can save thousands of lives,” it added.
How PMSMA Works
The government noted that PMSMA works through:
- Early identification of high-risk pregnancies
- Regular monitoring
- Timely medical intervention
"PMSMA has demonstrated that when quality healthcare reaches women at the right time, it saves lives, prevents complications and creates healthier beginnings for families and future generations," the govt said.
Services Offered Under PMSMA
The PMSMA service package includes clinical examinations, laboratory investigations (blood and urine tests), ultrasonography, medicines, and counselling on nutrition, birth planning, and safe pregnancy practices.
Key features include:
- Minimum one comprehensive and quality antenatal check-up by an Obstetrician and Gynecologist or a Comprehensive Emergency Obstetric and Newborn Care (CEmONC) trained doctor during the second or third trimester.
- Mobilizing pregnant women for specialist antenatal care services at designated public health facilities.
- High-risk pregnancy identification by screening for 25 high-risk factors and management at an early stage.
- Linking high-risk pregnancies to the nearest First Referral Unit (FRU) for safe delivery.
- Empanelment of private service providers for PMSMA service provision.
- Essential blood and urine investigations and ultrasonography.
- Counselling on nutrition, appropriate birth planning, and complication readiness for every pregnant woman.
Who Can Access PMSMA Services?
The program is available to:
- Pregnant women in their second trimester (13–27 weeks) or third trimester (28 weeks until delivery).
- Indian citizens residing in India who are willing to visit designated government health facilities on the 9th of every month.
- High-risk pregnancy cases, which receive priority attention and follow-up care.
- Women who are dropouts from regular antenatal care and are actively encouraged to participate.
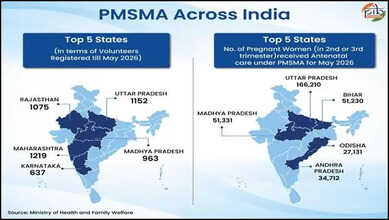
What Is Maternal Mortality Ratio?
MMR is defined as the number of maternal deaths per one lakh live births during a given period.
A maternal death refers to the death of a woman during pregnancy or within 42 days of termination of pregnancy due to causes related to or aggravated by the pregnancy or its management, excluding accidental or incidental causes.
Maternal health remains one of the clearest indicators of the strength and inclusiveness of a country's healthcare system.
"As India marches towards Viksit Bharat@2047, PMSMA will continue to transform the maternal healthcare landscape by ensuring that every pregnancy is safer and every mother receives the care she deserves. With continued commitment and collective action, PMSMA can help usher in an era where no woman loses her life while giving life, strengthening the foundations of a healthier and more prosperous India," the statement said.
Brazil Pauses Butantan Dengue Vaccine As Precaution After Two Deaths
Credits: Canva
Brazil has decided to temporarily suspend the world's first-ever dengue vaccine after two deaths. The jab, Butantan-DV, was stalled by Brazil's Ministry of Health on Monday. The authorities have reported that the decision came as a preventive step; it does not conclude that the deaths were connected with the shot.
The ministry has also given information that about 500 thousand doses of vaccine already administered by 30th May 2026, and among them 3,703 reports of adverse effects, thus it is roughly 0.7% of those vaccinated. Among these 42 cases, one was severe dengue and caused abdominal pain, persistent vomiting, and bleeding; thus, this group represents about 0.008% of the total immunized.
The National Pharmacovigilance Committee recommended this suspension. Analyzing the situation and studying reports made by municipal, state, and federal surveillance systems.
According to the Minister of Health, Alexandre Padilha, three cases were classified as severe. Of this total, two resulted in death.
As per the reports, the Butantan Institute is going to convene a committee of experts for epidemiological investigation. The institute will be responsible for sharing data with the health authorities. According to Butantan, the vaccine has a global efficacy of 79.6% and 89% protection against severe dengue in a study published in the scientific journal Nature.
The Butantan-DV Vaccine
The Butantan-DV vaccine is tetravalent and offers protection against the four known serotypes: DENV-1, DENV-2, DENV-3, and DENV-4.
The vaccine uses live viruses that have been “weakened” (attenuated) in a laboratory.
Once administered, the vaccine controls replication of these attenuated viruses in the body -- a process which induces the immune system to produce neutralizing antibodies specific to each of the four serotypes.
The vaccines create immunity specific to each serotype to enable the body to recognize and neutralize each variant individually.
The Butantan-DV vaccine was approved by the Brazilian Health Regulatory Agency (ANVISA) on November 26, 2025, for use by the Brazilian population aged 12 to 59.
The country's Ministry of Health has incorporated the vaccine into the national immunization program in January
launched a pilot project to immunize 90 percent of the target population in states with a high burden
rolled out vaccination of primary care health professionals in February.
Also Read: Salmonella Outbreak: Instant Noodles Sicken Over 80 In The UK, Europe
What Is Dengue?
Dengue is a viral infection that is primarily transmitted to the human body through mosquito bites. About 100 to 400 million people across the globe get infected with this disease every year. Usually, this condition is asymptomatic in its initial stages and leads to mild illness. However, occasionally, the virus can develop severe dengue infections that can even cause the death of the infected person.
© 2024 Bennett, Coleman & Company Limited

